In connection with the registration process of medical devices in Indonesia, where at the initial step Applicants isrequired to determine the risk classification of medical devices independently, MoH found out that still many applicantsdetermine the class of medical devices incorrectly. The factors include:
- 1. Applicant does not have enough knowledge about the device being to be registered.
- 2. The complexity of the medical devices that are being registered.
- 3. Complexity of the system to determine classification of health devices which is not user friendly and is not easyto understand.
While doing classification incorrectly may resulting in several issues such as: affect a risk of patient safety, excessgovernment payment due to applicant choose higher classification that it should be, and will slow down the licensing time ofmedical devices.
Based on the description above, MOH made a new feature on the MOH website namely SIKLARA (Sistem Klasifikasi ResikoAlat Kesehatan) which is in English, Medical Device Risk Classification system. This feature aims to facilitate applicants inclassifying medical devices and minimalize errors in class determination.
SIKLARA can be accessed via a computer or smartphone. The ways to access SIKLARA are as follows:
- 1. Open www.Regalkes.depkes.go.id
- 2. On the information menu click SIKLARA
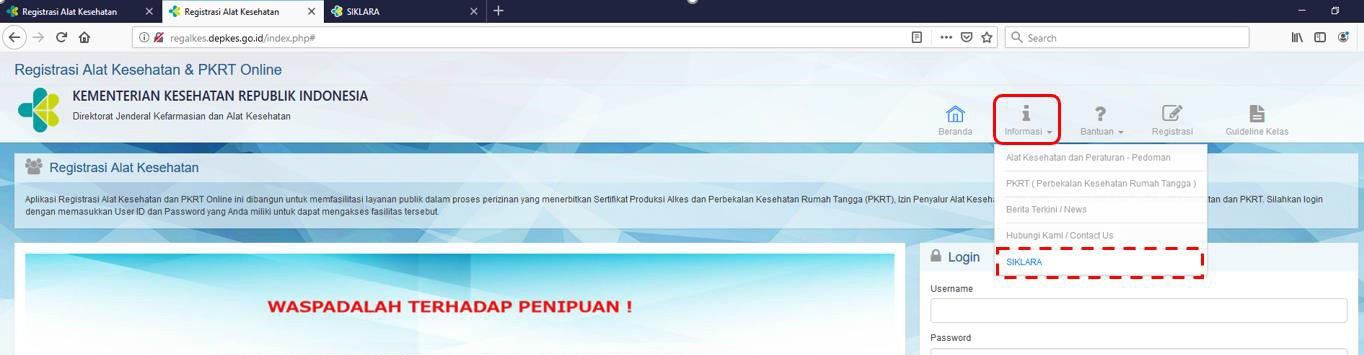
3. Once clicked, the SIKLARA page will appear as follows
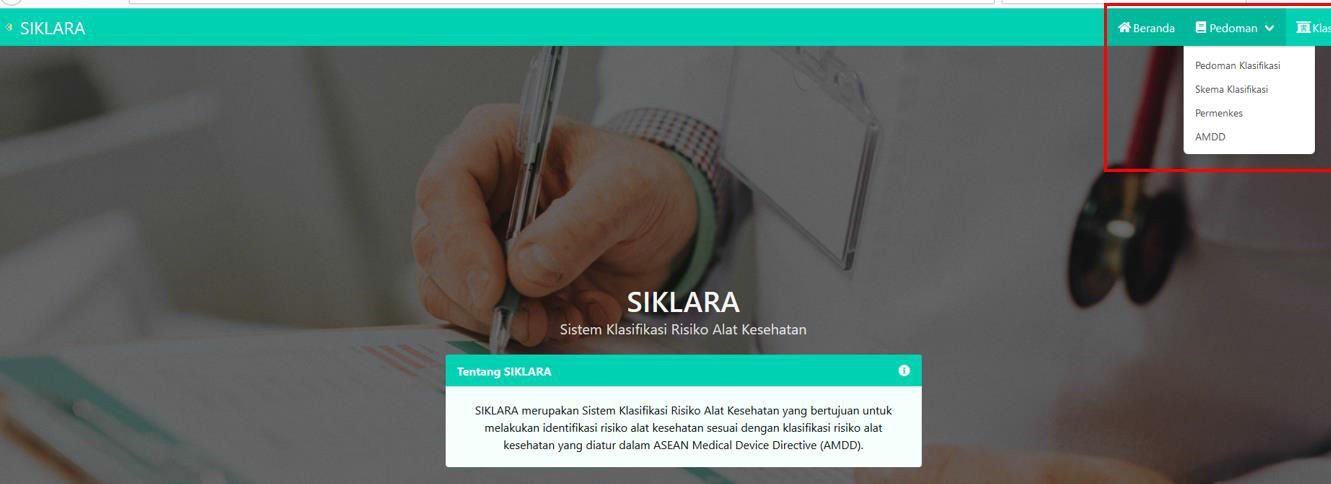
The features of SIKLARA include:
- a. Beranda (Home)
- b. Pedoman (Guidance)
- - Pedoman Klasifikasi (Classification Guidance)
- - Skema Klasifikasi (Scheme of Classification)
- - Permenkes (Ministry of Health Regulation)
- AMDD
- c. Klasifikasi (Classification)
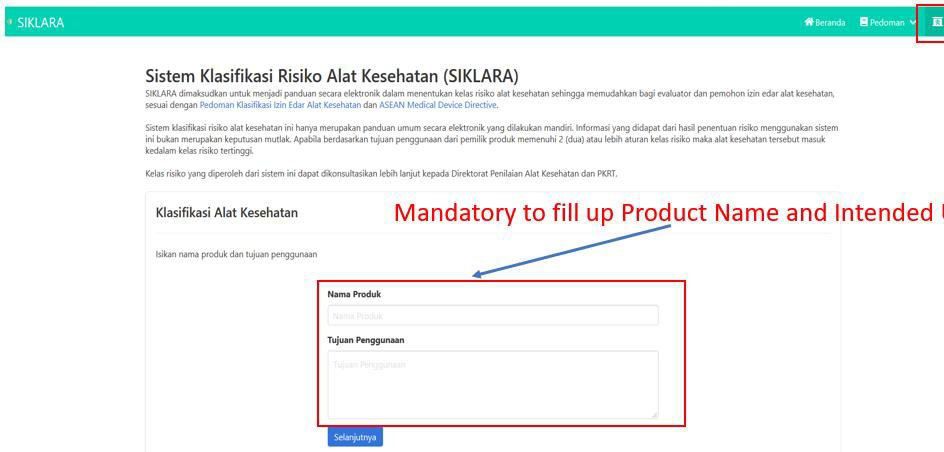
- 4. Click Classification Menu, and Classification page will be appeared. Fill up the product name and intended use and clickNext.
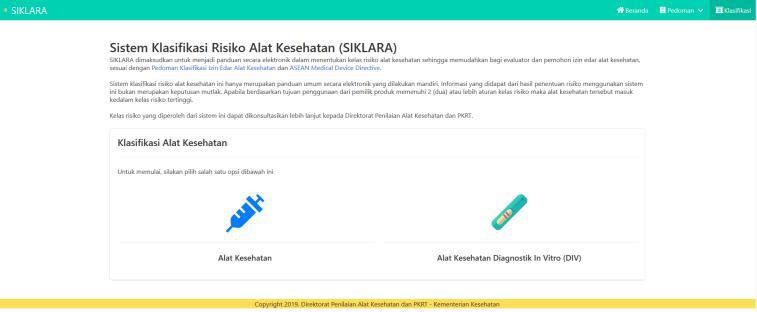
- 5. Start to answer according to the question
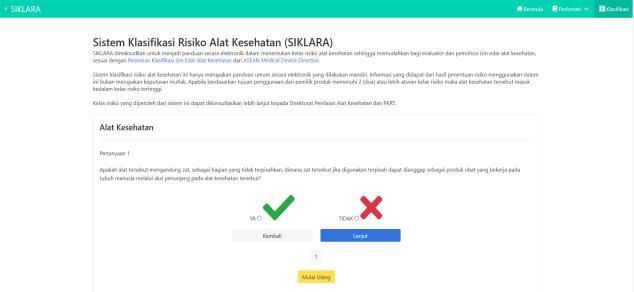
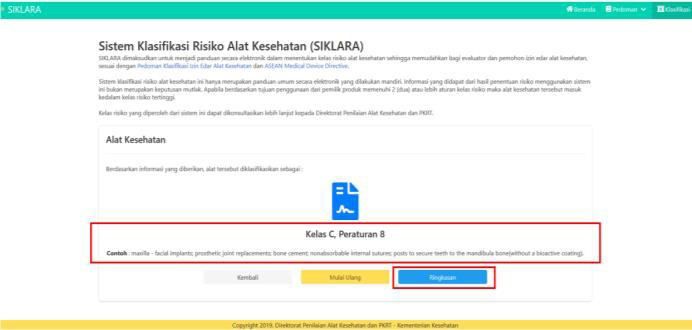
- 6. After completing answering questions on the system, the system will issue the classification results. The resultsthat will appear are Class, Rule and Sample Device.
-
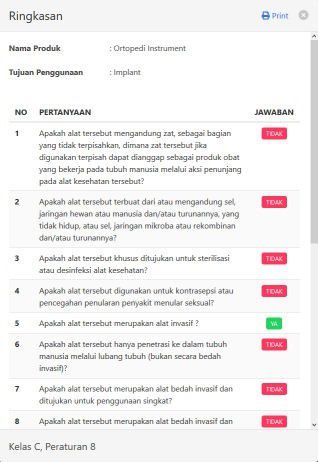
In addition, the applicant can also see and print a summary of questions and answers, where the Printable Resultscan be used as proof of self assessment and can be consulted with the Directorate of Medical Device Assessmentbecause this medical risk classification system is only an electronic guide that is carried out independently andinformation obtained from the results of determining the risk of using this system is not an absolute decision.
-
However, this classification system is still not linked to online registration system, where usually the applicantdetermine the class based on the category, sub category and product type of the devices which follow US FDA. MoH hasn’tmentioned the target on when to synchronize SIKLARA with online registration system yet.
-
-
