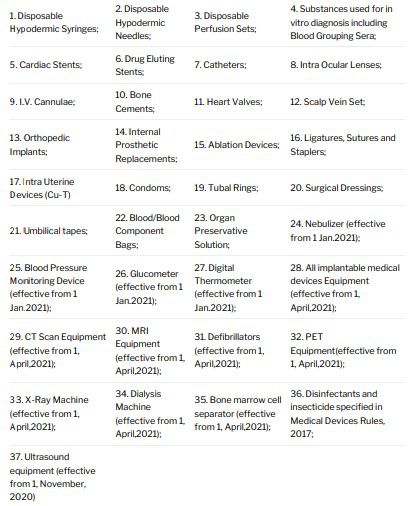
Until February 11, 2020, the Government had regulated or notified 37 categories of medical devices as drugs,
On February 11, 2020, the government exercised its powers to notify one or more categories of medical devices as “drug” to actually notify a new definition of medical devices. As per the notification, effective April 1, 2020, the medical devices that fall under the following definition will be regulated as “drug” under the Drugs and Cosmetics Act, 1940 and Medical Devices Rules, 2017:
All devices including an instrument, apparatus, appliance, implant, material or other article, whether used alone or in combination, including a software or an accessory, intended by its manufacturer to be used specially for human beings or animals which does not achieve the primary intended action in or on human body or animals by any pharmacological or immunological or metabolic means, but which may assist in its intended function by such means for one or more of the specific purposes of
- Diagnosis, prevention, monitoring, treatment or alleviation of any disease or disorder;
- Diagnosis, monitoring, treatment, alleviation or assistance for, any injury or disability;
- Investigation, replacement or modification or support of the anatomy or of a physiological process;
- Supporting or sustaining life;
- Disinfection of medical devices; and
- Control of conception.
The above new definition is intended to cover all medical devices, as per technical discussions that preceded the notification of the above definition. Thus, by virtue of this definition, all medical devices sold in India will come to be regulated by DCA and MDR from April 1, 2020, when the definition takes effect.
