Regulatory Background
Obsolete or discontinued medical devices that are still in use in healthcare facilities, such as hospitals, will be granted specific exemptions under the Medical Device (Exemption) Order 2024. This is because certain categories of these medical device may not be able to meet mandatory registration requirements. To control these situations, the Medical Device Authority (MDA) has published the First Edition of the Guidance Document on the Application for Confirmation Status of Obsolete and Discontinued Medical Device (MDA/GD/0071). This guidance was developed to assist establishments and healthcare professionals in understanding the applicable regulatory framework and in implementing appropriate control measures for these types of device categories.
Criteria for Obsolete and Discontinued Medical Devices
The medical device will be categorized as an obsolete and discontinued medical device if the device meets the criteria and category. The criteria for the application of confirmation of the medical device status are as follows:
| No | Status | Criteria |
| 1. | Obsolete medical device |
1. The medical device was previously registered with the MDA but can no longer be registered or have its registration maintained because the legal manufacturer has declared it obsolete, and the necessary technical documents are no longer available for registration purposes; or 2. The production of accessories has ceased or has been declared discontinued; and 3. The medical device is still in use in the facility* and in well-functioned and maintained condition. Note: Healthcare facilities should inform the establishment if they are using obsolete medical devices |
| 2. | Discontinued medical device |
1. The medical device was previously registered with the MDA but can no longer be registered or have its registration maintained because the legal manufacturer or AR has stopped the distribution in Malaysia; and 2. The medical device is still in use in the facility* and in well-functioned and maintained condition. Note: Healthcare facilities should inform the establishment if they are using discontinued medical devices |
Application Process and Requirements
The application shall be made by the manufacturer or AR by using the link form provided in the guidance. The process is summarized in Annex A in Figure 1. The application shall be accompanied by supporting documents as follows:
- Declaration letter from Legal manufacturer (Annex C);
- Declaration letter from the healthcare facility agreeing to use the device (Annex D);
- Estimated timeline or transition plan for maintenance support
- Completed and signed Attestation & Declaration Form with official company stamp (Annex E)
Applicants must complete, sign, and stamp all declarations. Upon submission, an acknowledgement email will be issued to the applicant.
Fees and Timeline
A RM300 processing charge applies per application and will not be refunded in cases of rejection or withdrawal. After submission, the payment invoice will be issued within 1–7 working days through the BayarNow platform, and applicants will be notified by email once it is ready. Payment must be made within 3 working days from the date the invoice is issued. The application will be considered withdrawn if the payment is not made within this period. The turn-around time per application is 10 working days upon submission of a complete application received and payment. A Letter of Exemption will be issued for the medical devices that are confirmed to be under obsolete or discontinued. The letter will remain valid for three (3) years. No extension will be granted beyond this validity period. A new application is required to be made if the medical devices continued to be used after the letter expiration date.
Post-Approval Obligations
Applicants must comply with several obligations for medical devices confirmed as obsolete or discontinued:.
- The devices cannot be imported or distributed
- Applicants must continue providing technical assistance, maintenance support, and full post-market responsibilities until the end of the device's lifespan.
- All related records such as distribution, complaints, Mandatory Problem Reporting (MPR), Field Corrective Actions (FCA), and recall documents must be retained for five (5) years from the date of End of Support (EOS) and must be provided to the Authority upon request.
MDA may instruct users to discontinue using the device if it is found unsafe or ineffective.
Clarification Through Visual
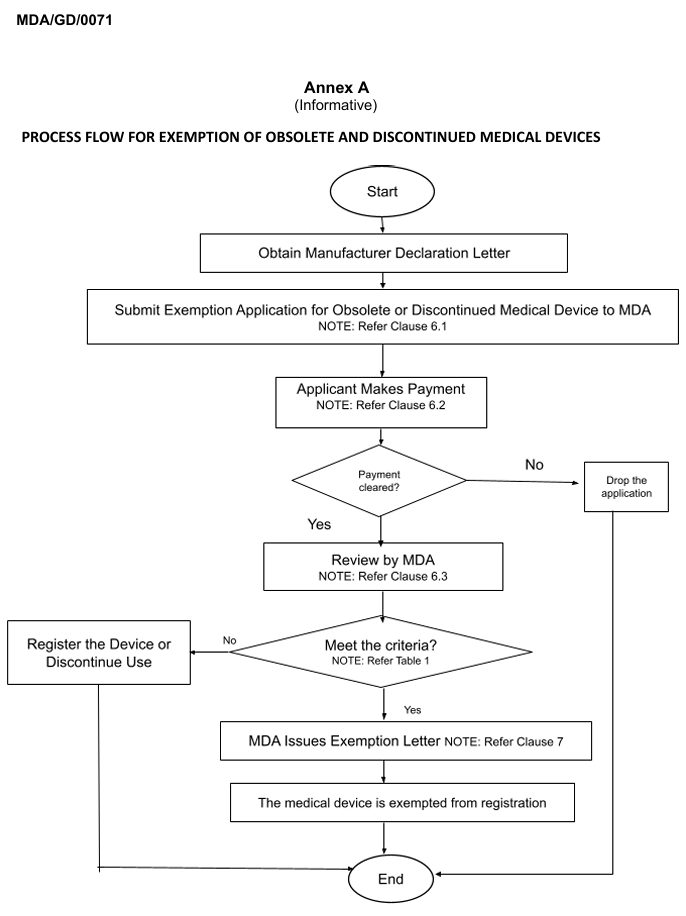
Companies managing legacy medical devices in Malaysia should assess whether their products fall under obsolete or discontinued categories and ensure timely application for exemption.
Qualtech supports manufacturers and Authorized Representatives in preparing exemption applications and managing post-market obligations in compliance with Medical Device Authority (MDA) requirements.
Contact us to ensure continued compliance and proper lifecycle management of your medical devices in Malaysia.
Reference
